American Journal of Veterinary Research, 87(3), November, 2025
Federica Gesuete, DVM1* ; Filippo Ferri, DVM, DECVIM2; Sara Muñoz Declara, DVM, DECVD1; Luca Luciani, DVM3; Federica Tirrito, DVM, DECVN4,5,6; Giordana Zanna, DVM, PhD, DECVD1
1 Department of Dermatology, Anicura Istituto Veterinario di Novara, SP9, Granozzo con Monticello, Italy
2 Department of Internal Medicine, Anicura Istituto Veterinario di Novara, SP9, Granozzo con Monticello, Italy
3 Department of Dermatology, Anicura Ospedale Veterinario I Portoni Rossi, Via Roma, 57/A, Zola Predosa, Italy
4 Department of Neurology, Anicura Istituto Veterinario di Novara, Granozzo con Monticello, Italy
5 University of Bari “Aldo Moro”, Strada provinciale per Casamassima Km 3, Valenzano, Italy
6 Università degli Studi di Milano, Via dell’Università 6, Lodi, Italy
*Corresponding author: Dr. Gesuete ([email protected])
Abstract
Objective
To describe the clinical effects of photobiomodulation (PBM) therapy using a dual-wavelength near-infrared laser (Multiwave Locked System; Asa Srl) as adjunctive treatment in dogs with calcinosis cutis (CC).
Animals
3 client-owned dogs presented to 2 referral veterinary hospitals from 2022 through 2023 were included. Cases were identified retrospectively via medical record review. Inclusion criteria were clinical and cytological features consistent with CC and histopathological examination when available, diagnosis of hyperadrenocorticism (HAC) based on clinical signs and diagnostic workup, availability of sufficient clinical records and photographic documentation, and owner consent for PBM therapy. Photobiomodulation was applied following a standardized protocol in scanning mode with a 2-cm collimated handpiece (approx 3-cm2 spot): 3 sessions during week 1, every 72 hours in week 2, and then weekly with 4 J/cm2 at the first session and 2.03 J/cm2 thereafter.
Clinical Presentation
Included dogs were a 5-year-old male Labrador Retriever and an 8-year-old male English Bulldog with naturally occurring HAC and a 3-year-old female German Shepherd with iatrogenic HAC. All presented with dermatologic lesions compatible with CC (erythematous-crusted plaques, erosive-ulcerative lesions, and/or papules).
Results
2 dogs achieved complete lesion resolution with hair regrowth within 7 to 12 weeks; the third showed partial improvement. No adverse events occurred.
Clinical Relevance
Findings, although based on a limited number of cases, suggest a potential supportive role for PBM in the management of CC. Controlled studies are warranted to establish standardized treatment parameters and clarify its role in CC.
Calcinosis cutis (CC) is an uncommon condition characterized by the deposition of inorganic, insoluble minerals in the dermis, subcutis, or, rarely, epidermis. It generally involves the accumulation of cations (ie, calcium) and anions (ie, phosphate or carbonate), which are typically in equilibrium in the extracellular fluid within the collagen fibers and elastin of the dermis.1,2
Based on their etiology, 4 subtypes of CC have been described: dystrophic, metastatic, idiopathic, and iatrogenic. Dystrophic is the most common form of CC, often associated with hyperadrenocorticism (HAC), either naturally occurring or iatrogenic.1–3 Inflammation and hypoxia are believed to contribute to tissue damage, acting as a focal point for dystrophic calcification.4 Despite this, there is limited evidence of effective treatments for CC in veterinary medicine.5 Recently, fluorescent light energy biomodulation, a form of photobiomodulation (PBM) consisting of a topical component containing specific chromophores that require activation by a light-emitting diode lamp, has been demonstrated to be useful in managing pyoderma associated with CC in a dog.6
In general, experimental and clinical studies suggest that PBM may beneficially influence biological tissues by reducing inflammation and improving tissue microcirculation.7–8 Furthermore, some in vitro and in vivo studies9–10 have reported potential effects on bacterial growth, although evidence in veterinary clinical settings remains limited.
This case series describes the effectiveness of PBM using a high-power, dual-wavelength near-infrared (NIR) laser source (Multiwavelength Locked System [MLS] laser; ASA Srl) as adjunctive therapy in producing a regression of skin lesions associated with CC in 3 dogs.
Methods
Case Selection
This descriptive case series included 3 client-owned dogs evaluated for dermatological lesions consistent with CC from 2022 through 2023. Cases were identified retrospectively by reviewing the medical records of 2 veterinary facilities in Italy: AniCura Istituto Veterinario di Novara (Granozzo con Monticello, Novara; cases 1 and 2) and AniCura Portoni Rossi (Zola Predosa, Bologna; case 3).
Inclusion criteria were (1) clinical presentation compatible with CC supported by cytological or histopathological findings when available, (2) diagnosis of HAC, (3) availability of sufficient clinical records and photographic documentation, and (4) owner consent for adjunctive PBM therapy. Exclusion criteria included insufficient medical documentation or use of a nonstandardized PBM protocol.
All procedures were performed with owner informed consent and in compliance with patient welfare. No sedation was required for diagnostic or therapeutic procedures.
Photobiomodulation therapy was standardized across cases using a high-power, dual-wavelength NIR laser with 2 different wavelengths within the NIR spectrum: 808 nm in continuous or frequency-modulated mode and 905 nm in pulsed mode. Treatments were applied in scanning mode with a 2-cm collimated handpiece (approx 3-cm2 spot size). Each affected area was divided into multiple sections to ensure uniform dosing. The first session delivered 4 J/cm2 (“infected wound” mode), whereas subsequent sessions delivered 2.03 J/cm2 (“wound” mode). Sessions were performed 3 times during week 1, every 72 hours during week 2, and then weekly thereafter. Cases 1 and 3 received PBM for approximately 12 weeks and case 2 for 7 weeks.
Concomitant therapies
Dogs received standard management for HAC. Other therapies included topical antiseptics and systemic or topical antimicrobials when cytology confirmed secondary superficial pyoderma. Routes, dosages, and administration frequencies are reported in the case descriptions.
Safety considerations
Protective goggles were worn by the operator and handlers throughout all PBM sessions. Treatments were performed without sedation, respecting patient behavior and welfare. No adverse events were observed during therapy.
Follow-up
Monitoring consisted of serial clinical examinations and photographic documentation. Follow-up duration was determined by lesion resolution or until PBM treatment was discontinued.
Statistical analysis
No formal statistical analyses were performed due to the descriptive nature and limited number of cases. Data were compiled and summarized using Excel 365 (Excel 365; Microsoft Corp, Version 16.89) to organize clinical records, images, and follow-up information.
Results
A total of 3 dogs were identified during the study period, and none were excluded after eligibility screening. The diagnostic approach varied among cases, reflecting real-world clinical practice.
Case 1
A 5-year-old intact male Labrador Retriever was referred with a 1-month history of obtunded mental status and decreased appetite. The owners reported polyuria and polydipsia as well as episodes of epistaxis over the previous few months. On physical examination, the patient showed marked lethargy, diffuse cutaneous lesions, and an increased body condition score of 7/9. Dermatological examination revealed the presence of 2 symmetrical erythematous, crusted-plaque lesions on the temporal region; a large, crusted plaque on the neck; and alopecia associated with 2 erythematous, crusted plaques across the dorsal thorax and dorso-lumbar region. Microscopic examination of plucked hairs showed hair shafts in the telogen phase. Multiple skin scrapings were negative for ectoparasites. Cytology of the plaques revealed a variable number of macrophages and neutrophils as well as the presence of extracellular and intracellular cocci and the presence of amorphous crystalline deposits.
Complete bloodwork and serum biochemistry showed mild lymphopenia (0.99 K/μL; reference range, 1.06 to 4.95 K/μL), marked increases in ALP (1,800 U/L; reference range, 14 to 147 U/L), and moderately elevated ALT (327 U/L; reference range, 25 to 122 U/L,), GGT (31 U/L; reference range, 2 to 13 U/L), and glutamate dehydrogenase (56 U/L; reference range, 1 to 18 U/L). Urine specific gravity was 1,022, with a urinary protein-to-creatinine ratio of 2.42 (reference range, < 0.5). Abdominal ultrasound revealed bilateral adrenomegaly and hepatomegaly, with the liver being diffusely hyperechoic.
Based on these results, naturally occurring HAC with secondary CC was suspected. To confirm the diagnosis, the following tests were recommended: the low-dose dexamethasone suppression test and skin biopsy under local anesthesia for dermatopathological examination. Additionally, to rule out infectious diseases, bacteriological and mycological examinations from deep skin tissue were also performed. The low-dose dexamethasone suppression test showed a lack of suppression at any time points (before: 3.7 µg/dL, range 1.0 to 6.0 µg/dL; 4 hours after: 3.5 µg/dL, range 0 to 1.4 µg/dL; 8 hours after: 2.7, range 0 to 1.4 µg/dL).
Skin biopsy specimens were collected from the plaque lesions on the neck and on the dorso-lumbar region. Histological examination showed severe and diffuse orthokeratotic hyperkeratosis of the epidermis, with mild-to-moderate irregular hyperplasia. Multifocal deposits of amorphous, crystalline, and deeply basophilic material were observed in the mid- and deep dermis, consistent with dermal mineralization. These mineralized structures were surrounded by a marked accumulation of activated fibroblasts and macrophages and occasionally accompanied by multinucleated giant cells. Both bacteriological and mycological examinations from deep skin tissue were negative.
To investigate the abnormal mental status, and considering the results of the previous examinations, an MRI scan (1.5 Tesla; SIGNA Creator; General Electric) of the head was performed. The scan revealed a large space-occupying lesion (measuring 26 X 26 X 24 mm) in the sellar region, which extended dorsally and exerted clinically important compression on the surrounding brain structures, including the optic chiasm, hypothalamus, and thalamus. The pituitary-to-brain ratio was > 0.31. The MRI scan findings were primarily compatible with a pituitary tumor, and a pituitary macroadenoma was suspected. A final diagnosis of pituitary-dependent hypercortisolism, due to pituitary macroadenoma, with cutaneous CC and secondary superficial bacterial infection, was proposed.
Treatment with trilostane (Vetoryl) was administered orally at a dose of 0.7 mg/kg every 12 hours, resulting in a rapid improvement of clinical signs. Additional treatments, such as radiation therapy and cabergoline, were declined by the owners. Due to the extensive skin lesions, topical therapy with chlorhexidine and systemic antibiotic therapy with amoxicillin-clavulanic acid (Synulox) administered orally at a dose of 20 mg/kg twice daily were started.
Photobiomodulation therapy was proposed as an additional treatment to address the dermatological lesions caused by mineral deposition, following the protocol in the flowchart (Figure 1). Owner informed consent was obtained, and all procedures were based on respecting patient behavior and welfare. The operator wore protective goggles throughout the procedure to ensure safety. After 14 days of initiating PBM therapy, antibiotic treatment was discontinued as serial cytology evaluations confirmed the improvement of superficial pyoderma. From that point onward, the only treatments were PBM therapy and topical chlorhexidine.
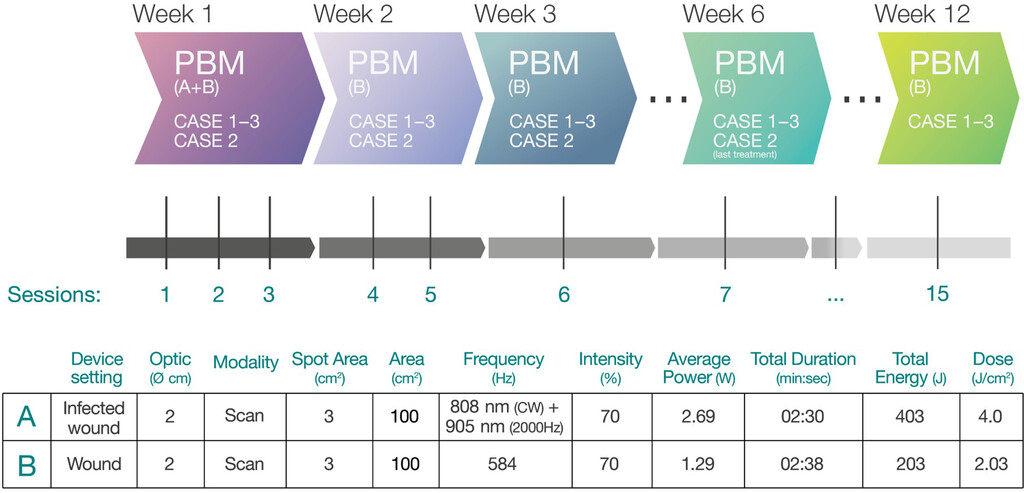
A class IV laser (Multiwave Locked System; Asa Srl) was used in all 3 cases. Three sessions were performed during the first week, followed by treatments every 72 hours in the second week and weekly sessions thereafter. The treatment was conducted in scanning mode using a collimated-lens handpiece (2-cm diameter, 3-cm2 spot area). In cases 1 and 3, sessions continued for a total of 12 weeks, whereas treatments were conducted for a total of 6 weeks in case 2. The operator wore protective goggles during all sessions to ensure safety. The affected areas were divided into multiple sections, with each section receiving the same dosage of 4 J/cm2 (“infected wound” mode) during the first session, followed by a dosage of 2.03 J/cm2 (“wound” mode) in subsequent sessions. The flowchart illustrates the different settings using a reference area of 100 cm2.
Three months later, there was significant improvement in the dermatological lesions, with visible hair regrowth (Figures 2 and 3). Photobiomodulation therapy was then discontinued.
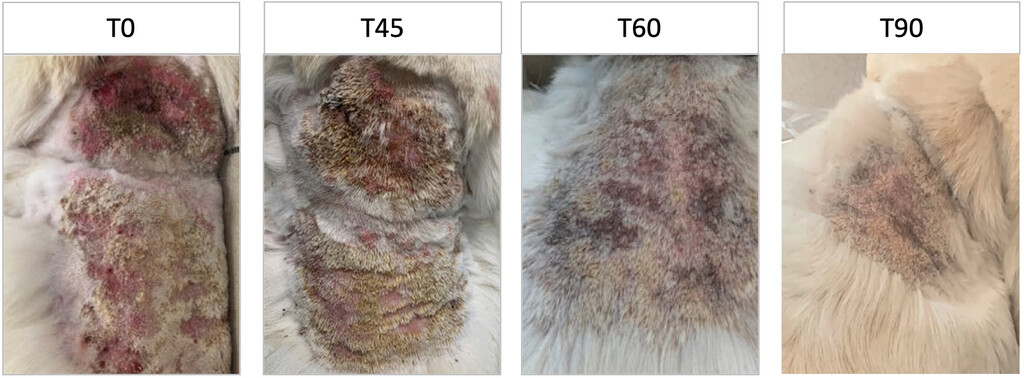
Wound healing progression of crusted plaque-like lesions on the neck of case 1, a 5-year-old male Labrador Retriever, treated with adjunctive PBM therapy using a dual-wavelength near-infrared laser (808 and 905 nm; Multiwave Locked System; Asa Srl). Images were taken at time (T)-0 (baseline), T45, T60, and T90 days. At T0, lesions exhibit crusting and marked erythema. By T45 and T60, crusting and inflammation are visibly reduced. At T90, lesions are resolved and hair regrowth is evident. These images illustrate the progressive effect of PBM on lesion resolution and tissue repair. Study period: 2022 through 2023.
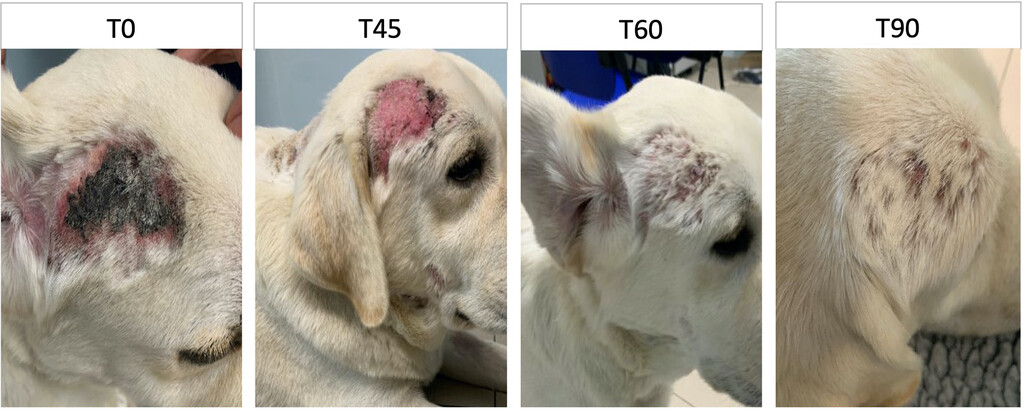
Wound healing progression in the right temporal region of case 1, a 5-year-old male Labrador Retriever, treated with adjunctive PBM therapy using a dual-wavelength near-infrared laser (808 and 905 nm; Multiwave Locked System; Asa Srl). Images were taken at different time points during treatment: T0, T45, T60, and T90 days. At T0, the temporal area shows crusts, marked erythema, and inflammation. By T45, crusts are reduced while erythema and inflammation persist. At T60, inflammation is markedly reduced, with initial hair regrowth visible. At T90, healing is complete, with full hair regrowth and a normal appearance of the treated area. The images illustrate the gradual and effective impact of PBM on tissue repair and regeneration in this case. Study period: 2022 through 2023.
Six months after diagnosis, the dog was reevaluated for anorexia and worsening of neurological signs, presenting disorientation, head pressing, circling, and pain upon palpation of the cervical column. A pain treatment with tramadol (3 mg/kg) and paracetamol (15 mg/kg) was started, but 2 weeks later the owner opted for euthanasia due to the poor evolution of the dog’s clinical conditions.
Case 2
A 3-year-old intact female German Shepherd was referred for a 2-week history of an erosive-ulcerative lesion on the neck. Topical treatment with streptomycin (q 12 h) was initially prescribed by the referring veterinarian for 10 days, but no improvement was observed. The dog had a previous history of primary immune-mediated nonerosive polyarthritis and meningomyelitis of unknown origin diagnosed through a comprehensive workup, including a total body CT scan, fine-needle aspiration cytology of multiple joints, and an MRI study of the vertebral column with cerebrospinal fluid analysis. Blood screening for vector-borne diseases (including Ehrlichia spp, Leishmania spp, Borrelia burgdorferi, Babesia canis, Anaplasma spp, Dirofilaria immitis, and Hepatozoon spp) ruled out infectious diseases.
After the initial diagnosis of immune-mediated disease, the dog had been receiving prednisolone therapy (1.5 mg/kg once daily) for 2 months.
At the time of presentation to the dermatology service, the immune-mediated condition appeared to be in remission. The owners reported that, following glucocorticoid therapy, the dog had developed severe polyuria, polydipsia, and polyphagia.
During physical examination, the dog exhibited mild lethargy, a distended abdomen, and generalized muscle atrophy. Dermatological evaluation revealed an 8 X 8-cm erosive-ulcerative lesion on the dorsal neck, along with 2 firm, white-coated lesions on the lateral tongue and perilabial region.
The collateral diagnostics performed included microscopic examination of plucked hairs, which showed follicles predominantly in the telogen phase, and multiple superficial and deep skin scrapings, which were negative for parasites. Cytology of the erosive-ulcerative lesions demonstrated abundant amorphous, basophilic material with a few crystal-like structures, neutrophils, macrophages, multinucleated giant cells, and extracellular cocci.
Blood analyses were performed and revealed marked increases in ALT (1,125 U/L), ALP (1,081 U/L), and glutamate dehydrogenase (110 U/L), with mild elevations in AST (117 U/L) and GGT (46 U/L). Further diagnostic tests, including dermatopathology, bacteriology, and mycology examination, were proposed but declined by the owner.
Based on the clinical history, cytological findings, and bloodwork, a diagnosis of CC secondary to iatrogenic HAC, complicated by a superficial bacterial infection, was made.
Glucocorticoid therapy was gradually tapered and discontinued over a 4-week period to minimize the relapse risk of the immune-mediated disease. Topical gentamicin (q 12 h) was initiated, and PBM therapy was applied exclusively to the dorsal neck lesion according to the protocol outlined in Figure 1.
Owner informed consent was obtained, and all procedures respected patient welfare. The operator wore protective goggles throughout treatment. Topical therapy was discontinued after 10 days, following serial cytological evaluations showing progressive improvement of superficial pyoderma. After 25 days of treatment, an > 80% reduction in lesion size was observed, and complete clinical resolution of the dorsal neck lesion was achieved 7 weeks after initiation of laser therapy.
Case 3
An 8-year-old intact male English Bulldog was referred for a 2-month history of progressive polyuria and polydipsia and dermatological lesions. On physical examination, the dog was panting, and a distended abdomen was noted. Dermatological examination revealed erythematous plaques with crusts on the dorsal neck region along with several erythematous papules on the left shoulder area, some of which were confluent and crusted.
Microscopic examination of plucked hairs revealed a predominantly telogen phase. Multiple superficial-to-deep skin scrapings were negative for ectoparasites. Cytological examination of the lesions revealed a mixture of neutrophils, macrophages, and a few multinucleated giant cells around some crystal-like structures. Additionally, cocci were observed, with both intracellular and extracellular distribution. The CBC revealed an increased platelet count (761 X 103/μL; reference range, 150 to 500 X 103/μL) and mild lymphopenia (800 K/μL; reference range, 900 to 4,000 K/μL). Serum biochemistry showed elevated ALP (551 U/L; reference range, 14 to 147 U/L), mildly increased ALT (113 U/L; reference range, 15 to 65 U/L) and GGT (16.7 U/L; reference range, 2 to 13 U/L). Urine specific gravity was 1,004, with a urinary protein-to-creatinine ratio of 12.22 (range, < 0.5).
The ACTH stimulation test (pre-ACTH cortisol: 8.4 µg/dL, range 1 to 4.8 µg/dL; post-ACTH cortisol: 30.80 µg/dL, range 6 to 18 µg/dL) was positive for hypercortisolism. A total body CT scan revealed a 7.3 X 13 X 8-mm pituitary mass in the sellar region, extending dorsally to the basisphenoid, with a pituitary-to-brain ratio > 0.31, hepatomegaly, and bilateral adrenomegaly. A pituitary macroadenoma was considered the most likely diagnosis. The owner declined other diagnostic investigations, such as dermatopathological, bacteriological, and/or mycological examinations of deep skin tissue.
Based on the patient’s history, the type and distribution of the lesions, and the results of the investigations, a diagnosis of macroadenoma with secondary CC and secondary bacterial infection was made.
Treatment with trilostane (Vetoryl; 0.6 mg/kg, q 12 h) was started; to treat the CC and the concurrent secondary bacterial infection, topical therapy with chlorhexidine and systemic therapy with amoxicillin-clavulanic acid (Synulox; 20 mg/kg twice daily) were started. Photobiomodulation therapy was also introduced following the protocol in the flowchart (Figure 1). After 10 days of combined therapy, the antibiotic treatment was discontinued as the pyoderma improved. Owner informed consent was obtained, and all procedures were based on respecting patient behavior and welfare. The operator wore protective goggles throughout the procedure to ensure safety. Clinical improvement of the dermatological clinical signs was observed from the eighth PBM session, with significant progress noted by the sixth week. However, some lesions remained partially improved and persisted at week 12, and complete resolution was not achieved.
Three months after the initial presentation, the dog developed severe dyspnea and 2 episodes of collapse; an acute pulmonary thromboembolism was suspected. The owners declined further diagnostic tests, and compassionate euthanasia was performed.
Discussion
To the best of the authors’ knowledge, this is the first report to describe the use of MLS for the treatment of CC in 2 cases with naturally occurring HAC and in 1 with iatrogenic HAC.
There are no broadly effective treatments for CC owing to the lack of a complete understanding of the mechanisms involved. In human medicine, a variety of treatment modalities are used based on the type, severity, and number of lesions. These include pharmacological therapies, such as antibiotics, immunosuppressants, bisphosphonates, colchicine, IV immunoglobulins, and other biologic therapies. Additionally, nonpharmacological treatments, such as surgical excision, shockwave therapy, and carbon dioxide laser therapy, are also employed.11 Similarly, in veterinary medicine there is no standardization of therapies for CC, and many treatments are supported by limited case reports or rely on the clinical expertise of practitioners. For example, there is one case report12 that describes a dog with CC locally treated with DMSO with a resolution of wounds in 3 months. Dimethyl sulfoxide is known for its high skin-penetrating and anti-inflammatory properties, but its efficacy remains anecdotal as do the mode and frequency of application. The effectiveness and safety of minocycline, a bacteriostatic antibiotic in the tetracycline family used for CC in human medicine, has also been assessed in 5 dogs, with a clinical resolution observed in 4 of 5 cases in 6 to 12 weeks. It is thought that minocycline reduces inflammation, chelates calcium and iron, and influences angiogenesis beyond its antibacterial properties.5 Finally, an accelerated improvement in lesions treated with fluorescent light energy compared to those not exposed and treated with DMSO and chlorhexidine was recently documented in a dog with CC.6
In general, PBM is a nonthermal process that utilizes endogenous chromophores to promote photophysical and photochemical reactions at various biological levels.13 This process involves light in the red or NIR spectrum, with specific wavelengths ranging from 600 to 1,100 nm. Lasers, light-emitting diodes, and various other light-emitting devices with class 3B and class 4 lasers are commonly employed as they emit monochromatic, coherent, and collimated light, enabling precise and targeted treatment of specific tissues.14 Although the exact mechanism of PBM is not fully understood, it could depend on the target and type of cell being modulated. It is highly likely that photons are absorbed by cytochrome c oxidase, which is in the inner membrane of mitochondria. Cytochrome c oxidase absorbs light in the range of 500 to 1,100 nm, becomes activated, and dissociates from nitric oxide, binding to oxygen. This results in improved electron transport and ATP production and the triggering of several pathways that lead to cell proliferation and tissue repair.15,16 On this basis, the use of PBM has been reported for the management of musculoskeletal disorders and neurological diseases, to relieve pain, and, especially, for treatment of wounds given its noninvasive nature and its minimal side effects in both human and veterinary medicine.17–25
However, the efficacy of PBM on target tissues also depends on treatment parameters, light source, wavelength, density, type of energy emission (continued or pulsed), type of lasers, and duration and frequency of laser application.26 Although there have been several in vivo studies,25 results in veterinary medicine are inconclusive due to the lack of standardization of parameters under different clinical conditions. In this case series, we introduced a more standardized approach by following a specific protocol.
The laser used was the MLS laser, a class 4 therapy system emitting NIR beams at 2 wavelengths within the NIR spectrum: 808 nm in continuous or frequency-modulated mode and 905 nm in pulsed mode. The beams are spatially overlapped and synchronized during treatment. The laser has an average maximum power of 3.5 W and a peak power of 270 W.
With the treatments detailed here, complete remission of CC was achieved in 2 of the 3 cases in 7 to 12 weeks. In the case associated with iatrogenic HAC, lesions resolved after approximately 7 weeks—shorter than the 12 weeks reported by Huang et al27 following corticosteroid withdrawal alone. In the dog affected by naturally occurring HAC, lesion resolution occurred after 12 weeks, consistent with previously reported cases treated with minocycline or DMSO. It is likely that additional treatment sessions in the third case would have resulted in complete clinical resolution.
It was hypothesized that these therapeutic effects could be due to multifactorial mechanisms. Based on previously reported effects of PBM, it is possible that laser therapy contributed to modulating inflammation7–29 and supporting tissue repair, enhancing microcirculation,30 reducing local edema,31 and promoting a more favorable environment for healing and resorption of calcium deposits.
Second, the superficial bacterial infections commonly associated with CC lesions also play a critical role in the disease progression, often complicating the healing process. The potential antibacterial effect of PBM,32 along with its role in impacting the skin microbiome as recently documented,33 may have contributed to reducing the bacterial load and shortening the duration of antibiotic use, thereby promoting tissue regeneration and helping to alleviate inflammation.
These observations suggest that PBM may have contributed to accelerated lesion healing. However, due to the small number of cases and the absence of controls, its specific contribution to the clinical outcome cannot be determined. While these findings are descriptive and cannot establish efficacy, PBM may represent a promising adjunctive approach for managing CC in dogs, warranting further investigation in larger, controlled studies.
Acknowledgments: None reported.
Disclosures: The authors have nothing to disclose. No AI-assisted technologies were used in the composition of this manuscript.
Funding: The authors have nothing to disclose.
ORCID:
Federica Gesuete https://orcid.org/0009-0002-4253-1494
Luca Luciani https://orcid.org/0000-0002-5294-5116
References
- Gross TL, Ihrke PJ. Degenerative, dysplastic and depositional diseases of dermal connective tissue. In: Gross TL, Ihrke PJ, eds. Skin Diseases of the Dog and Cat: Clinical and Histopathologic Diagnosis. 2nd ed. Blackwell Science Ltd; 2005:373–377.
- Doerr KA, Outerbridge CA, White SD, et al. Calcinosis cutis in dogs: histopathological and clinical analysis of 46 cases. Vet Dermatol. 2013;24(3):355–379. doi:10.1111/vde.12026
- Miller WH Jr, Griffin CE, Campbell KL. Neoplastic and nonneoplastic tumors. In: Miller WH Jr, Griffin CE, Campbell KL, eds. Muller and Kirk’s Small Animal Dermatology. 7th ed. Elsevier; 2013:831–832.
- Valenzuela A, Chung L. Management of calcinosis associated with systemic sclerosis. Curr Treat Options Rheumatol. 2016;2:85–96. doi:10.1007/s40674-016-0035-x
- Jang HJ, Kang MH, Sur JC, Park HM. Minocycline as a treatment of dog with calcinosis cutis. Korean J Vet Res. 2013;53(4):253–256. doi:10.14405/kjvr.2013.53.4.253
- Apostolopoulos N, Mayer U. Use of fluorescent light energy for the management of bacterial skin infection associated with canine calcinosis cutis lesions. Vet Rec Case Rep. 2020;8(4):e001285. doi:10.1136/vetreccr-2020-001285
- Hamblin MR. Mechanisms and applications of the anti-inflammatory effects of photobiomodulation. AIMS Biophys. 2017;4(3):337–361. doi:10.3934/biophy.2017.3.337
- Zhang G, Yi L, Wang C, et al. Photobiomodulation promotes angiogenesis in wound healing through stimulating the nuclear translocation of VEGFR2 and STAT3. J Photochem Photobiol B. 2022;237:112573. doi:10.1016/j.jphotobiol.2022.112573
- de Sousa NT, Gomes RC, Santos MF, Brandino HE, Martinez R, de Jesus Guirro RR. Red and infrared laser therapy inhibits in vitro growth of major bacterial species that commonly colonize skin ulcers. Lasers Med Sci. 2016;31(3):549–556. doi:10.1007/s10103-016-1907-x
- Rico-Holgado S, Ortiz-Díez G, Martín-Espada MC, Fernández-Pérez C, Baquero-Artigao MR, SuárezRedondo M. Effect of low-level laser therapy on bacterial counts of contaminated traumatic wounds in dogs. J Lasers Med Sci. 2021;12:e78. doi:10.34172/jlms.2021.78
- Le C, Bedocs PM. Calcinosis cutis. In: StatPearls. StatPearls Publishing; 2023.
- Tolon JMC, Jimenez JJE, Gilabert II, Trasobares PC. Resolution of iatrogenic calcinosis cutis in a dog through topical application of DMSO. Vet Rec Case Rep. 2018;6(3):e000619. doi:10.1136/vetreccr-2018-0006
- Anders JJ, Lanzafame RJ, Arany PR. Low-level light/laser therapy versus photobiomodulation therapy. Photomed Laser Surg. 2015;33(4):183–184. doi:10.1089/pho.2015.9848
- Fesseha H. Laser therapy and its potential application in veterinary practice—a review. J Light Laser Curr Trends. 2020;3:007.
- Glass GE. Photobiomodulation: a review of the molecular evidence for low level light therapy. J Plast Reconstr Aesthet Surg. 2021;74(5):1050–1060. doi:10.1016/j.bjps. 2020.12.059
- de Freitas LF, Hamblin MR. Proposed mechanisms of photobiomodulation or low-level light therapy. IEEE J Sel Top Quantum Electron. 2016;22(3):7000417. doi:10.1109/JSTQE.2016.2561201
- DE Oliveira MF, Johnson DS, Demchak T, Tomazoni SS, Leal-Junior EC. Low-intensity LASER and LED (photobiomodulation therapy) for pain control of the most common musculoskeletal conditions. Eur J Phys Rehabil Med. 2022;58(2):282–289. doi:10.23736/S1973-9087.21.07236-1
- Farazi N, Salehi-Pourmehr H, Farajdokht F, Mahmoudi J, Sadigh-Eteghad S. Photobiomodulation combination therapy as a new insight in neurological disorders: a comprehensive systematic review. BMC Neurol. 2024;24(1):101.
doi:10.1186/s12883-024-03593-4 - González-Muñoz A, Cuevas-Cervera M, Pérez-Montilla JJ, et al. Efficacy of photobiomodulation therapy in the treatment of pain and inflammation: a literature review. Healthcare (Basel). 2023;11(7):938. doi:10.3390/
healthcare11070938 - Pavlov SB, Babenko NM, Kumetchko MV, Litvinova OB, Mikhaylusov RN. Experimental study of the effect of photobiomodulation therapy on the regulation of the healing process of chronic wounds. Int J Photoenergy. 2021;2021(1):3947895. doi:10.1155/2021/3947895
- Hochman L. Photobiomodulation therapy in veterinary medicine: a review. Top Companion Anim Med. 2018;33(3): 83–88. doi:10.1053/j.tcam.2018.06.004
- Miller LA, Alves JC. Musculoskeletal disorders and osteoarthritis. In: Winkler CJ, Miller LA, eds. Laser Therapy in Veterinary Medicine: Photobiomodulation. John Wiley & Sons Inc; 2025:129–150. doi:10.1002/9781394205356.ch11
- Hernández-Bule ML, Naharro-Rodríguez J, Bacci S, Fernández-Guarino M. Unlocking the power of light on the skin: a comprehensive review on photobiomodulation. Int J Mol Sci. 2024;25(8):4483. doi:10.3390/ijms25084483
- Pryor B, Millis DL. Therapeutic laser in veterinary medicine. Vet Clin North Am Small Anim Pract. 2015;45(1): 45–56. doi:10.1016/j.cvsm.2014.09.003
- Millis DL, Bergh A. A systematic literature review of complementary and alternative veterinary medicine: laser therapy. Animals (Basel). 2023;13(4):667. doi:10.3390/ani13040667
- Lima PLV, Pereira CV, Nissanka N, et al. Photobiomodulation enhancement of cell proliferation at 660 nm does not require cytochrome c oxidase. J Photochem Photobiol B. 2019;194:71–75. doi:10.1016/j.jphotobiol.2019.03.015
- Huang HP, Yang HL, Liang SL, Lien YH, Chen KY. Iatrogenic hyperadrenocorticism in 28 dogs. J Am Anim Hosp Assoc. 1999;35(3):200–207. doi:10.5326/15473317-35-3-200
- Salman S, Guermonprez C, Peno-Mazzarino L, et al. Photobiomodulation controls keratinocytes inflammatory response through Nrf2 and reduces Langerhans cells activation. Antioxidants. 2023;12(3):766. doi:10.3390/antiox12030766
- Shamloo S, Defensor E, Ciari P, et al. The anti-inflammatory effects of photobiomodulation are mediated by cytokines: evidence from a mouse model of inflammation. Front Neurosci. 2023;17:1150156. doi:10.3389/fnins.2023.1150156
- Joniová J, Bechelli C, Déglise S, Allagnat F, Wagnières G. Stimulating angiogenesis and post-ischemic tissue repair: optimizing photobiomodulation parameters in vivo. J Photochem Photobiol B. 2025;269:113202. doi:10.1016/j.jphotobiol.2025.113202
- Albertini R, Villaverde AB, Aimbire F, et al. Anti-inflammatory effects of low-level laser therapy (LLLT) with two different red wavelengths (660 nm and 684 nm) in carrageenan-induced rat paw edema. J Photochem Photobiol B. 2007;89(1):50–55. doi:10.1016/j.jphotobiol.2007.08.005
- Amaroli A, Ravera S, Zekiy A, Benedicenti S, Pasquale CA. A narrative review on oral and periodontal bacteria microbiota photobiomodulation, through visible and nearinfrared light: from the origins to modern therapies. Int J Mol Sci. 2022;23(3):1372. doi:10.3390/ijms23031372
- Muñoz Declara S, D’Alessandro A, Gori A, et al. Evaluation of the impact of near-infrared Multiwavelength Locked System laser therapy on skin microbiome in atopic dogs. Animals. 2024;14(6):906. doi:10.3390/ani14060906



