More than 20 million people in the United States are estimated to have some form of peripheral neuropathy.1
Diabetes is the leading cause of chronic peripheral neuropathy. An estimated 60 to 70 percent of people with diabetes will experience damage to sensory, motor, or automatic nerves.1 As the prevalence of diabetes continues to rise, so do the complications associated with the disease.2
Damage to sensory nerves can lead to loss of sensation or an inability to feel pain, increased pain and hypersensitivity, and/or difficulty with coordination and balance. Peripheral sensory neuropathy can lead to ulcers, infection, gangrene, and amputation of the foot and/or leg, especially in cases caused by diabetes.1
The most common approach to treating neuropathic pain is through prescription medications. However, less than half of patients report satisfactory relief of their symptoms.3-5 Additionally, these pharmaceuticals can be associated with harmful side-effects or lead to drug dependence. The ineffectiveness and potential risks of these drugs calls for a new solution.5
While research and treatment is often aimed at symptomatic relief, treatments that attempt to heal injured peripheral nerves with minimal side effects are needed.2
In recent years, laser therapy has gained recognition as a viable nonpharmacological approach to relieve neuropathic pain while promoting the regeneration and proliferation of peripheral nerves.5,6
Laser Therapy
Laser therapy uses an induced emission of non-ionizing electromagnetic radiation to transfer energy to biological cells to stimulate cellular activity and biological processes.
A number of studies have shown that laser radiation is effective in improving cell energy metabolism by increasing ATP production.5 This starts a cascade of molecular reactions that improve cell function and enhance the body’s natural healing processes. Some of these effects can include:
- Increased tissue regeneration and wound healing by stimulating fibroblast development.
- Reduced inflammation and edema by stimulating blood flow and lymphatic drainage, and by interacting with the synthesis and degradation of inflammation mediators.
- Induced analgesia by stimulating the release of endorphins and enkephalins to block the transmission of pain sensations to the brain.7
Laser therapy can be used in the treatment of many musculoskeletal conditions.7 Studies also show that laser therapy can induce a biostimulatory effect on the nervous system by promoting microcirculation in the treated area, increasing nerve function, increasing axonal growth and myelinization, and improving the regeneration of peripheral nerves.8
These findings indicate that laser therapy can be an effective solution for reducing neuropathic pain while addressing an underlying cause and thus improving quality of life for patients. However, this efficacy is largely dependent on treatment protocols and parameters, including treatment frequency, duration, irradiation mode, wavelength, power, and pulse frequency.5
Innovations in Laser Therapy Technologies
Based on over 30 years of research, the Multiwave Locked System® (MLS) Therapy Laser uses a patented and FDA-cleared emission system to precisely synchronize two wavelengths of light energy (808 nm and 905 nm) using continuous and pulsed emission modes to provide optimal clinical effectiveness.
Through the synchronization of these emissions, the Multiwave Locked System pulse is able to transfer light energy to the cells more effectively leading to an intense and immediate therapeutic effect that is greater than either emission used individually or together in a non-synchronized manner. The MLS pulse also assures that energy will be delivered to the maximum possible tissue depth of up to 5 centimeters.7,9
MLS Laser Therapy has no known side effects and limited contraindications.7
Clinical Research
MLS Laser Therapy has been developed and tested following a strict course of biomedical and clinical research. The effectiveness of the synchronized emissions were initially tested in vitro, in vivo, and by means of controlled clinical trials run by state-of-the-art health organizations for treating traumatic and degenerative painful diseases.7
Italy’s University of Turin’s biology department as well as Brazil’s University of Padova’s departments of biology and anatomy have studied and verified that the clinical effectiveness of the MLS pulse provides more efficient biostimulatory results with less energy in considerably reduced times in comparison to traditional low level laser therapy.7
Effects on Inflammation
The continuous 808 nm emission decreases inflammation by stimulating blood flow and lymphatic drainage inducing the reabsorption of fluid buildup and by interacting with the synthesis and degradation of inflammation mediators.7
Studies show that MLS Laser Therapy irradiation induces an increase in NLRP 10, a protein that inhibits the activity of caspase 1 and PYCARD protein complex therefore inhibiting the production of the inflammatory cytokines interleukin to reduce inflammation.4
The decrease in inflammation leads to a normalization of vascular function and thus decreases edema. By reducing inflammation and edema, there is a decrease in painful symptoms.4
Some studies also suggest that laser therapy can decrease inflammation by means of microglia and macrophage activation.10
Macrophages and microglia have two different activated states that exhibit a range of responses to injury, including pro-inflammatory and anti-inflammatory actions. Injury to peripheral nerves cause these cells to release pro-inflammatory factors that can cause heightened sensitivity of nerves, as well as the disinhibition of pain modulation. These studies suggest that laser therapy can cause a shift in the activated state encouraging these cells to begin releasing anti-inflammatory factors instead, therefore decreasing mechanical hypersensitivity.10
Effects on Pain
While pain relief is felt as a secondary effect of reducing inflammation and edema, the MLS pulse also induces an analgesic effect. The pulsed 905 nm emission affects the transmission of pain at the level of superficial nociceptors and on afferent nervous fibers. This results in an increase of the cell and nervous fiber’s stimulation threshold and therefore a reduced sensation of pain.7
Moreover, the activation of the proprioceptive channels (A-alpha and A-beta fibers), which are subject to greater myelination and therefore able to conduct the nervous stimulus more quickly, block the transmission of painful sensations to the brain.7
The analgesic effect obtained is immediate and long lasting.7
Effects on Nerve Regeneration
Preliminary in vivo trials carried out on rats investigated the effects of laser therapy on nerve regeneration and the consequent restoration of muscle function following neurorrhaphy of the median and ulnar nerve. Results found a statistically significant faster recovery of muscle trophism and myelination of regenerating nerve fibers accompanied by a statistically significant faster recovery of the lesioned motor function in comparison to the placebo group.11
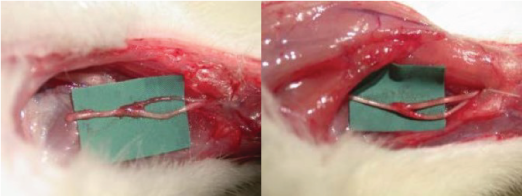
Right: Nerve Regeneration in Animal Treated with the combined MLS Pulse.11
In addition to assessing laser-treated vs non-lasers-treated animals, this study compared the synchronized MLS pulse with the individual continuous 808 nm and pulsed 905 nm emissions alone. While all emission types produced positive results, the best results in functional and muscle mass recovery were obtained by the MLS pulse showing that laser wavelength and dose are critical factors for the success of biostimulation.11
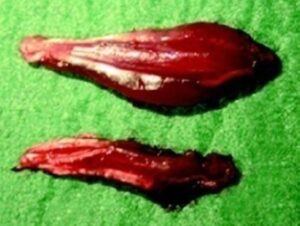
Further research investigating the role of MLS Laser Therapy in nerve regeneration observed significant improvements in functional, electrophysiological and morphometric measurements after nerve crushing in rats due to its anti-inflammatory and reparative effects.6
MLS Laser Therapy increases ATP production, nerve metabolism, and myelin production, and enhances axonal sprouting, thus increasing nerve proliferation while it decreases the retrograde degeneration of peripheral nerves and limits chromatolysis and neuronal atrophy after crushing.6
A separate study found that the MLS pulse has a beneficial effect on injured peripheral nerves and the neuropathic pain felt by patients diagnosed with diabetic peripheral neuropathy.2
Nerve conduction studies observing F-wave chronodispersion (Fc) measurements showed significant improvement in tibial and peroneal nerve function after MLS Laser Therapy treatment in patients with diabetic sensory neuropathy compared to untreated control nerves. Improvement in function will lead to improved sensibility in the feet therefore decreasing the likelihood of ulceration and infection.2
Effects on Wound Healing
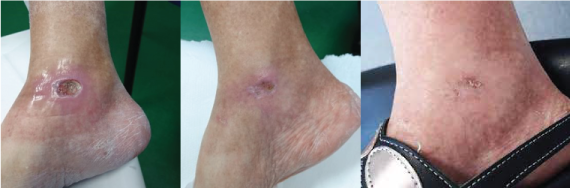
Some studies also indicate the potential for MLS Laser Therapy to assist in diabetic wound care.
Approximately 19 to 34% of individuals with diabetes will develop a diabetic foot ulcer in their lifetime, of which an estimated 20% will require lower extremity amputation and 10% will die within 1 year of their first diabetic foot ulcer diagnosis.12
Patients with diabetic foot ulcers are prone to infection and the healing process is complicated by diabetic neuropathy leading to chronic non-healing ulcers.
Research shows that the metabolic changes frequently found in diabetic wounds cause a prolonged inflammatory phase delaying granulation tissue formation. Studies investigating the efficacy of MLS Laser Therapy on chronic wound healing show promising results due to its anti-inflammatory and anti-edema effects and stimulation of tissue regeneration.13
Studies have revealed that MLS Laser Therapy increases modulation of several proteins involved in cell cycle regulation, cytoskeleton organization and differentiation indicating an anabolic cellular response.14
Studies also observed a significant increase in proteins able to bind ATP, which could be related to the increased availability of ATP. Enhanced anabolism and energy metabolism can better sustain the repair processes.14
A highly significant result is the increase of protein phosphatase-1 (PP1), a protein involved in many important biological processes including the regulation of glycogen metabolism and the process of relaxation and contraction in smooth muscles.14,15
Following the MLS treatment, total phosphatase activity increased by approximately 80%. These enzymes play a crucial role in biological functions and basic processes at the cellular level, including metabolism, gene transcription, cell cycle, cytoskeletal organization, stability and interaction of proteins, adhesion, migration and apoptosis. Migration and apoptosis are strongly involved in the healing process and regulate the succession of different cells populations at the wound site, including immune cells, endothelial cells and fibroblasts.15,16
Other studies show that MLS Laser Therapy can affect the production of molecules, such as collagen and metalloproteases, involved in extracellular matrix turnover, a fundamental step in the repair process.16
Final Considerations
As a novel but well-researched approach to managing pain and inflammation, laser therapy is filling the gap for non-pharmacological and non-invasive options for diabetic peripheral neuropathy and other nerve-related conditions.
With recent innovations to laser technologies, including the development of the MLS Laser Therapy emission system, therapy lasers are becoming more efficient in their therapeutic effects while remaining safe for the patient and intuitive to the operator.
Backed by thirty years of research, MLS Laser Therapy precisely synchronizes wavelengths to provide optimal clinical effectiveness offering an immediate analgesic response while promoting a strong anti-inflammatory action at the same time.
MLS Laser Therapy shows great potential as a viable approach to diabetic foot care. Since nerve inflammation plays an important role in the development and progression of neuropathy, the anti-inflammatory actions provoked by the MLS pulse are crucial to its effectiveness for managing neuropathic pain.
By supplementing traditional diabetic foot care protocols with laser therapy treatments, physicians have the opportunity to improve care and quality of life for patients by accelerating the body’s natural healing processes and reducing their reliance on pharmaceuticals that have limited effectiveness and possible side effects.
Learn More about the benefits of MLS Laser Therapy.